Nitrogen is the “silent workhorse” of the industrial world. Occupying roughly 78% of the Earth’s atmosphere, it is an inexhaustible resource that underpins everything from the food we eat to the smartphones we use. Because it is chemically inert, nitrogen is the primary tool used by engineers to combat the two most common enemies of industrial quality: Oxidation and Combustion.
As industries move toward “Industry 4.0,” the method of obtaining this gas has shifted from traditional liquid deliveries to sophisticated on-site generation. This guide explores why nitrogen is essential, how it is produced in nitrogen generator, and how to optimize its use for maximum efficiency.

1. The Chemical and Physical Nature of Nitrogen
To understand why nitrogen is used, one must understand its molecular structure. Nitrogen exists as a diatomic molecule N₂. The two nitrogen atoms are held together by a triple covalent bond, one of the strongest bonds in nature.
Key Properties:
Inertness: Unlike oxygen, which loves to react with metals (rust) and organic matter (spoiling), nitrogen is exceptionally stable.
Dryness: Industrially generated nitrogen is “bone dry,” meaning it has an extremely low dew point (often as low as -70°C). This makes it ideal for protecting moisture-sensitive electronics.
Non-Conductivity: It is an excellent dielectric gas, used in high-voltage applications.
2. On-Site Production Technologies
The most significant advancement in nitrogen utility is the ability to generate it on-demand. Below is the workflow of how ambient air is transformed into high-purity process gas.

A. Pressure Swing Adsorption (PSA)
PSA is the most common technology for high-purity requirements. It relies on the principle of adsorption, where gas molecules are trapped on the surface of a solid material.
The PSA Process Flow:
- Compression: Ambient air is compressed to roughly 7–10 bar.
- Purification: The air is filtered to remove oil, water, and dust.
- Adsorption (The Separation): The air enters a tower filled with Carbon Molecular Sieve (CMS). The CMS has pores of a specific size. Oxygen molecules (which are smaller than Nitrogen molecules) are trapped in these pores.
- Desorption: Once the CMS is full of oxygen, the pressure is dropped. The oxygen is “released” and vented to the atmosphere, while the purified nitrogen is sent to a storage tank.
Technical Note: PSA systems typically use a dual-tower design to ensure a continuous flow of gas; while one tower is adsorbing, the other is regenerating.
B. Membrane Separation Technology
If PSA is about “trapping,” Membrane technology is about “filtering.”
The Membrane Process Flow:
- Air Preparation: Compressed air is heated slightly to prevent condensation.
- Permeation: The air enters a bundle of thousands of hollow, hair-like fibers.
- The “Fast” vs. “Slow” Principle: Oxygen, $CO_2$, and water vapor are “fast” gases—they pass through the fiber walls easily. Nitrogen is a “slow” gas—it stays inside the fiber and travels all the way to the end.
- Collection: The nitrogen is collected at the outlet, while the waste gases are vented silently.

[Comparison Table: PSA vs. Membrane Nitrogen Generation]
| Feature | PSA Nitrogen Generator | Membrane Nitrogen Generator |
|---|---|---|
| Operating Principle | Carbon Molecular Sieve (CMS) adsorbs oxygen under high pressure. | Semi-permeable hollow fibers separate gases by permeation rates. |
| Nitrogen Purity | High to Ultra-High (99.9% – 99.999%). | Low to Medium (95% – 99.5%). |
| Key Advantage | Produces high-purity gas suitable for sensitive industrial processes. | Simple design, no moving parts, and nearly instantaneous start-up. |
| Maintenance | Moderate: Requires filter changes and monitoring of switching valves. | Very Low: Only requires pre-filtration maintenance. |
| Service Life | CMS lasts 10+ years if the feed air is kept clean and oil-free. | Membrane modules typically last 15–20 years with consistent use. |
| Capital Investment | Higher initial cost due to complex valves and control systems. | Lower initial cost; more economical for lower purity needs. |
| Noise Level | Higher (audible "puffing" sound during pressure release). | Very Low (quiet, continuous operation). |
| Environmental Fit | Stationary; requires more floor space and stable temperatures. | Compact, lightweight, and ideal for mobile or offshore applications. |
| Best For | Laser cutting, Electronics, Pharmaceuticals, Labs. | Tire inflation, Oil & Gas blanketing, Food packaging (MAP). |
3. Purity Standards and Their Industrial Applications
Not every application requires 99.999% purity. In fact, using a higher purity than necessary is a common waste of energy.
Case 1: Standard Purity (95% to 98%)
- Fire Suppression: In the mining and maritime industries, nitrogen is pumped into confined spaces to lower oxygen levels below 12%, making it impossible for a fire to start.
- Tire Inflation: In aviation and high-end automotive sectors, nitrogen is used because it lacks moisture (preventing rim rot) and expands/contracts less than oxygen under high heat.
Case 2: High Purity (99% to 99.9%)
- Food and Beverage (MAP): Modified Atmosphere Packaging is the process of flushing oxygen out of food containers (like salad bags or snack packs). This stops the “browning” of produce and the rancidity of fats.
- Coffee Encapsulation: To preserve the volatile aromas of ground coffee, nitrogen is used during the pod-filling process.
Case 3: Ultra-High Purity (99.99% to 99.999%)
- Electronics Manufacturing: During Reflow Soldering, nitrogen prevents the formation of “dross” (oxidized metal). Without it, modern micro-circuitry would fail due to poor connectivity.
- Laser Cutting: When cutting stainless steel, nitrogen acts as a shield gas. It blows away the molten metal so quickly that the heat doesn’t have time to burn the edges, resulting in a mirror-like finish.
4. Troubleshooting and Efficiency: The “Hidden” Costs
Maintaining a nitrogen system requires attention to three specific areas to prevent cost overruns:
I. The Air-to-Nitrogen Ratio
This is the most critical metric. For example, to get 1 unit of 99.9% nitrogen, you might need 3 units of compressed air. If you only need 98% purity, you might only need 2 units of air. Lowering your purity requirement by just 1% can often save 10–20% in energy costs.
II. Air Pre-Treatment
The biggest killer of nitrogen generators is oil and water contamination. If the air from the compressor isn’t properly dried and filtered before it hits the PSA or Membrane, the separation media will be “poisoned” and lose efficiency.
III. System Sizing
A system that is too small will cause pressure drops and purity fluctuations. A system that is too large will “cycle” too often, causing mechanical wear. Proper sizing involves calculating both the average flow and the peak flow of your facility.

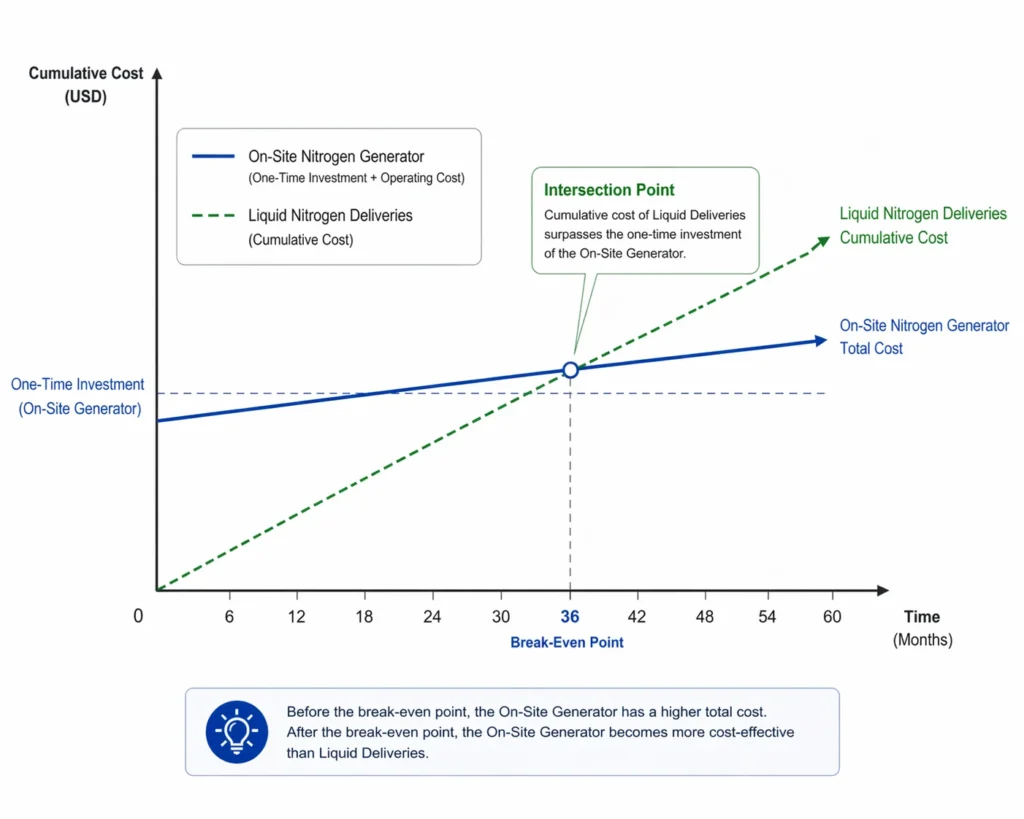
5. The Economic Shift: On-Site vs. Bulk Liquid
Historically, companies signed long-term contracts for liquid nitrogen (LIN) stored in massive cryogenic tanks. While convenient, this model has several flaws:
- Evaporation Loss: A liquid tank “bleeds” gas every day to prevent pressure buildup. This is known as Boil-off, and it can waste up to 10% of your purchased gas.
- The Carbon Footprint: Heavy trucks transporting cryogenic liquids contribute significantly to a company’s Scope 3 emissions.
- The Payback Period: Most on-site generators pay for themselves within 12 to 24 months, after which the nitrogen is essentially “free,” costing only the electricity used by the air compressor.
6. Future Trends: Nitrogen and Green Energy
As we look toward 2026 and beyond, nitrogen generators are becoming smarter. Modern systems are now equipped with IoT sensors that monitor purity and flow in real-time, adjusting the compressor speed to match demand. Furthermore, in the burgeoning Green Hydrogen industry, nitrogen is being used as a carrier gas and for safety purging in electrolysis plants, making it a key player in the global energy transition.
Conclusion
Nitrogen is more than just a gas; it is a critical safeguard for quality and safety across the global supply chain. By understanding the nuances between PSA and Membrane technologies and accurately defining purity needs, businesses can transition from being “gas consumers” to “gas producers.” This shift not only secures the supply chain but also drives significant bottom-line savings and environmental progress.
Showing Case of NItrogen Generator
FAQ
1. Why should I generate nitrogen on-site instead of buying liquid nitrogen?
On-site generation is typically 40% to 80% cheaper than liquid nitrogen deliveries. It eliminates long-term contracts, delivery surcharges, and “boil-off” (evaporation loss) from liquid tanks. Additionally, it reduces your carbon footprint by removing heavy truck deliveries from your supply chain.
2. What is the difference between Nitrogen Purity and Nitrogen Flow?
Purity refers to the percentage of nitrogen in the gas stream (e.g., 99.9%).
Flow refers to the volume of gas produced per hour (e.g., $m^3/h$ or SCFH).
It is important to note that as you increase the purity requirement on a generator, the available flow rate usually decreases.
3. Does a nitrogen generator require a lot of maintenance?
Not significantly. For PSA systems, maintenance involves checking valves and replacing air filters to protect the Carbon Molecular Sieve (CMS). For Membrane systems, maintenance is even lower, usually limited to changing the pre-filters to ensure no oil or water enters the membrane fibers.



